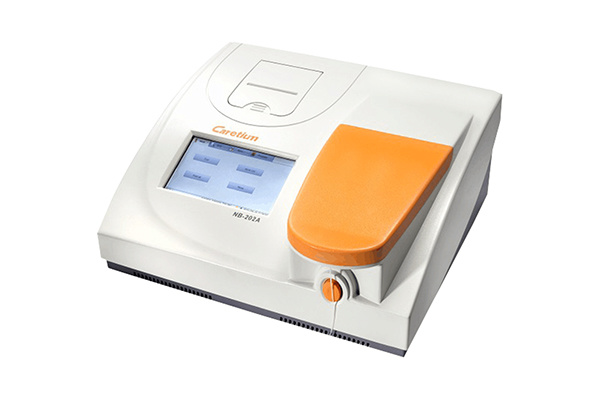
Caretium Medical Instruments Co., Limited was founded in 2001, a high-tech company focusing on the research and development, manufacturing, sales and after-sale service of in-vitro diagnostic equipment and reagents. Caretium has been certified as China’s national “High-tech Enterprise” from 2011, got CE mark, ISO 13485, ISO 9001, GMP certified by South Korea and other certifications.
All rights reserved:Caretium Medical Instruments Co., 粤ICP备11050458号 | Powered by :300.cn